A diagnosis of premature ovarian failure (POF) can be devastating for a person who wishes to start a family. The condition may not only limit their ability to conceive, but it also poses serious health risks, including ischemic heart disease.
Luckily, POF is rare. According to a study published in the journal Experimental and Therapeutic Medicine, the condition affects between 1 and 3 percent of women — although its incidence is rising. Careful ultrasound monitoring and a selection of emerging therapies offer new hope that patients with POF may achieve a healthy pregnancy.
Diagnosing Premature Ovarian Failure
The onset of POF may be sudden or gradual. In order to fit the prematurity standard, however, it must begin before age 40. Amenorrhea may be the first sign that prompts a patient to visit their OB/GYN. Although symptoms vary, a patient with undiagnosed POF may complain of heart palpitations and menopausal symptoms, including night sweats, irritability, insomnia, fatigue, anxiety, depression, vaginal dryness or a waning interest in sex.
The mechanisms behind premature ovarian failure are complex. The condition is associated with factors as diverse as damage from chemotherapy, endocrine disorders, infection and X chromosome abnormalities such as Turner syndrome. The etiology of POF may also be related to an autoimmune disorder, such as primary adrenal insufficiency.
Premature ovarian failure was once referred to as premature menopause. Today, its name and definition are still the subjects of lively debate.
POI vs. POF
Some clinicians use the labels premature ovarian failure and premature ovarian insufficiency (POI) interchangeably. Others distinguish based on prognosis, suggesting that the term "insufficiency" more accurately reflects the chance of restoring ovarian function with recent medical advances.
A 2018 paper from the Journal of Assisted Reproduction and Genetics suggests viewing the condition as a continuum, with premature ovarian failure landing on the extreme end of the spectrum. It cites earlier studies that characterize POF as occurring in women who are younger than 40, who have secondary amenorrhea that lasts at least four months and whose blood tests show postmenopausal levels of follicle-stimulating hormone (FSH).
However, FSH test results may be misleading in women whose ovulatory cycles intermittently swing from a normal to a postmenopausal range. A transvaginal ultrasound exam can be instrumental in confirming a POF diagnosis. For patients with premature ovarian failure, an ultrasound exam often reveals small ovaries with no sign of growing follicles.
Ultrasound also plays an important role in differential diagnosis by ruling out conditions that can cause primary or secondary amenorrhea, such as polycystic ovaries or anatomical abnormalities associated with Müllerian aplasia or Asherman syndrome.
Counting Antral Follicles in POF Diagnosis
An antral follicle count (AFC) is accepted as one of the best markers for assessing functional ovarian reserve. Clinicians routinely use high-resolution transvaginal ultrasound to evaluate AFC. This assessment is especially crucial for women with POF, many of whom have fewer than five to seven antral follicles in each ovary, according to the Journal of Assisted Reproduction and Genetics study.
SonoAVC™antral (Sonography-based Volume Calculation antral) allows a clinician to automatically count antral follicles in 3D in order to assess ovarian reserve in patients with suspected POF. After taking a volume of the whole ovary, this tool automatically segments follicles of 2 to 10 mm.
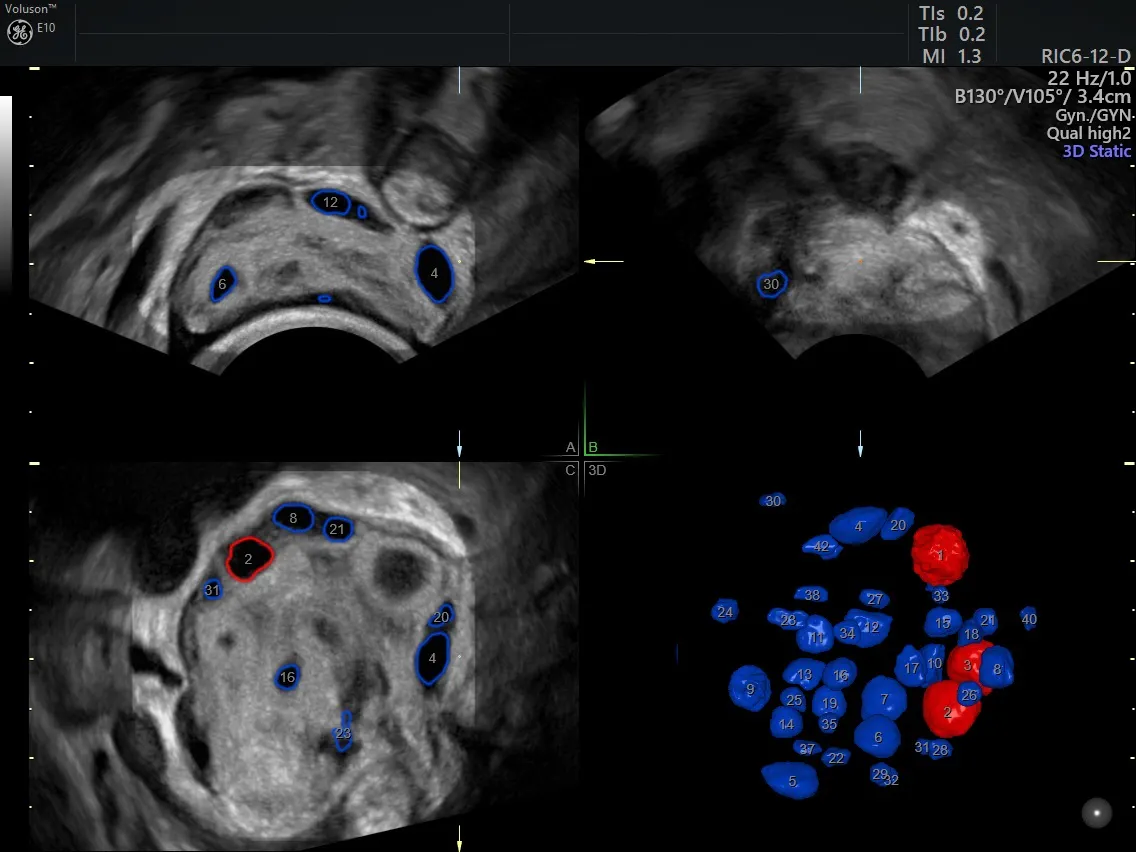
Automated antral follicle count with the SonoAVCantral Tool
Both clinicians and patients may find the counting process less stressful with SonoAVCantral. It allows volumes to be taken quickly, and the tool automatically segments and counts the antral follicles. Physicians can even run the software or review the volume after a patient has left the office.
Advances in POF Treatment
Hormone replacement therapy (HRT) is a first-line treatment to counteract estrogen deficiency in patients diagnosed with premature ovarian failure. Treatment choices include modified postmenopausal HRT regimes and oral contraceptives.
Historically, unassisted childbearing has been virtually impossible for the vast majority of people diagnosed with the condition. Estimates of spontaneous pregnancy rates among POF patients stand as low as 1.5 percent, according to a review published in Current Opinion in Obstetrics and Gynecology. For women who wanted to bear children, IVF and embryo transfer using donor eggs were previously the most effective options, according to the study.
However, current research is exploring the possibility of a POF patient conceiving with their own eggs. The Current Opinion in Obstetrics and Gynecology review describes a novel technique called in vitro activation, which has been shown to reactivate residual dormant follicles in patients with premature ovarian failure.
The technique involves harvesting ovarian tissue by removing one or both of the ovaries. The tissue is treated in vitro to stimulate follicle growth and then surgically reinserted beneath the fallopian tubes. Ultrasound monitoring, as well as monitoring of a patient's HCG and estradiol levels, is a critical part of postoperative follow-up. If follicle growth occurs, the oocytes can be retrieved and cultured in vitro until they reach the blastocyst stage and are ready to be implanted; alternatively, one study used intracytoplasmic sperm injection (ICSI). Thus far, in vitro activation has resulted in at least four pregnancies and two live births.
More recent studies have explored the use of stem cells to restore the ovarian structure and function in animal models of premature ovarian failure. A 2018 paper published in Reproductive Sciences found that injecting human bone marrow mesenchymal stem cells (BMSCs) in mice with chemotherapy-induced ovarian failure restarted the production of follicles, resulting in 12 pregnancies. The paper builds on earlier stem cell research in which women experiencing POF following chemotherapy achieved spontaneous pregnancies after a bone marrow transplant. While these results are still preliminary and have yet to be repeated in human patients, this treatment may show promise for those affected.