Are there barriers to treating vulvovaginal atrophy, also known as genitourinary syndrome of menopause (GSM)? Absolutely.
For one, its name implies that it only occurs as the result of menopause. Patients may think it is normal or are embarrassed to discuss their symptoms and do not seek help. Further, some physicians are unfamiliar with newer treatment options or are hesitant to prescribe hormones. Last but not least, treatment options can be expensive, messy and require motivation to use regularly.
Understanding Vulvovaginal Atrophy
Vulvovaginal atrophy has a variety of symptoms involving the lower genital tract, most commonly in postmenopausal women. However, it can also occur during the immediate postpartum stage, during extended periods of lactation, in premenopausal patients who smoke or are undergoing any treatments that cause a hypoestrogenic state.
Prolonged lowered estrogen levels may cause vulvovaginal dryness, burning, itching, irritation and vaginal discharge. The longer the duration of hypoestrogenism, the more severe the symptoms.
The urinary tract is also affected by lowered estrogen levels, causing symptoms such as frequency, dysuria, urethral discomfort, recurrent urinary tract infections (UTIs) and hematuria. Sexual activity can become painful due to decreased vaginal lubrication, leading to bleeding and decreased arousal, orgasm and sexual desire. The impact on quality of life can be significant.
Reaching a Vulvovaginal Atrophy Diagnosis
The annual well-woman visit is a prime opportunity to explore any urogenital or GSM problems a woman may be experiencing. Asking if there is a history of recurrent UTIs, vaginal discharge or bleeding with intercourse opens the door to topics some patients might otherwise omit. The exam should note any tissue fragility, fissures or a fused labia after menopause. Further GSM findings include vaginal color, presence or absence of rugae, and lubrication and urethral eversion or prolapse.
The diagnosis of vulvovaginal atrophy is clinical. A vaginal pH or maturation index may be helpful to evaluate treatment results but is not necessary. Testing for vaginitis and vaginosis and evaluating for dermatitis and skin conditions, such as lichen sclerosus or planus, with a vulvar biopsy should be considered on a case-by-case basis. For example, a woman with vulvar irritation who wears a panty liner daily to manage occasional stress incontinence may simply be experiencing discomfort from the combined exposure to the liner and urine, not GSM. Recommending pelvic floor physical therapy and avoiding the liner, rather than using estrogen treatments, may be the best solution.
Abnormal vaginal bleeding, especially of the postmenopausal variety, should always be investigated fully. A pelvic ultrasound can rule out uterine malignancy, the most common gynecologic cancer. In some, a Pap smear may be indicated. If the patient only notices blood when wiping after urinating, it may not be vaginal bleeding; other sources such as the bladder and rectum need to be examined. If the final diagnosis is GSM, a discussion regarding treatment options should begin.
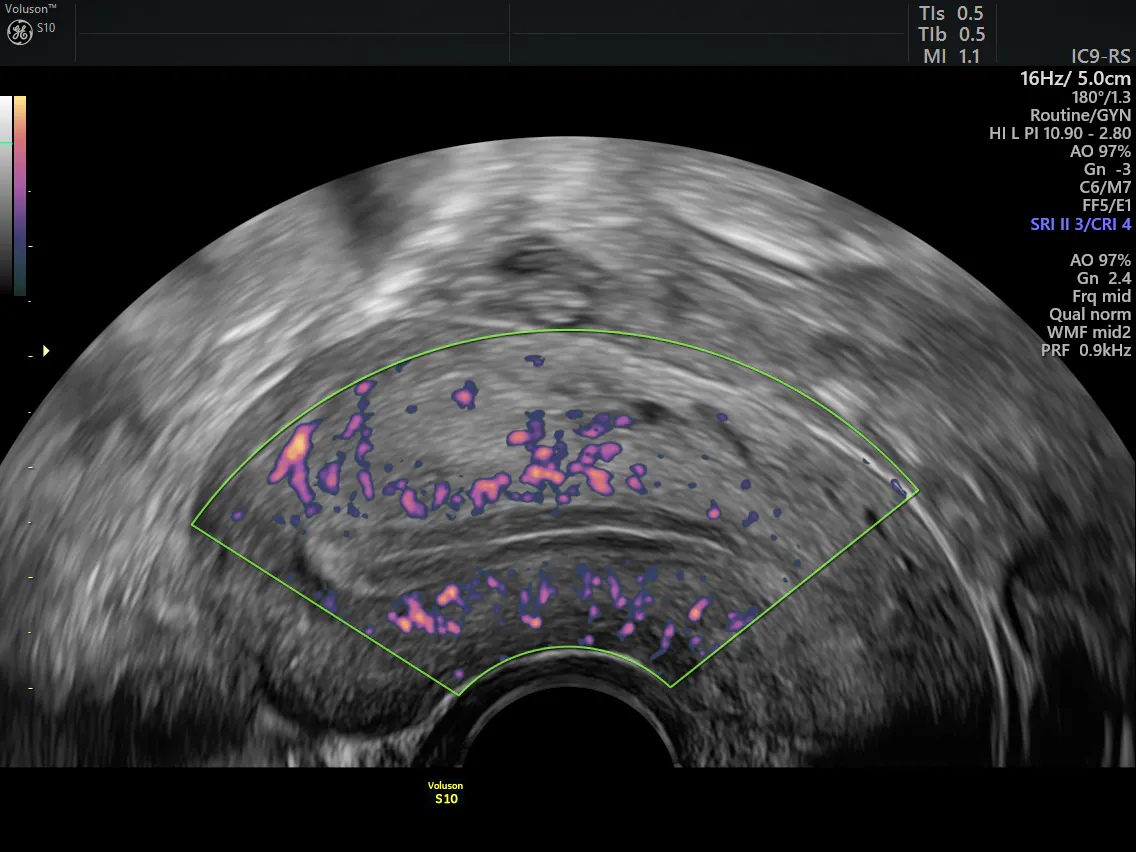
Transvaginal ultrasound of uterus
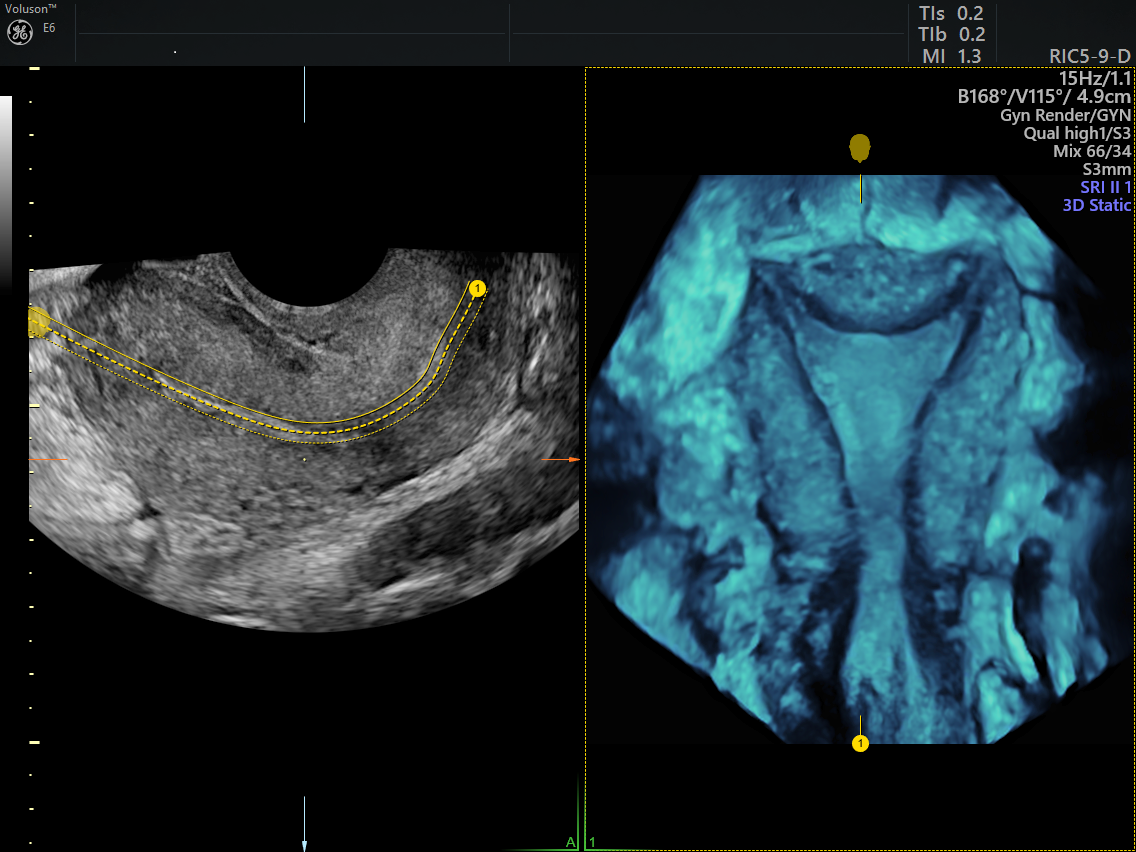
3D pelvic ultrasound with OmniView technology
Having a Conversation With Your Patients
Many providers are speaking about GSM with their patients prior to menopause or when prescribing medications that cause a hypoestrogenic state. The weekly use of over-the-counter vaginal moisturizers is helpful in maintaining moisture as the earliest symptoms develop. Personal lubricants that are water-, oil- or silicone-based can also be recommended for intimacy.
What works well for one patient does not always for the next. Today's options for treatment include a variety of delivery systems, including tablets, creams, vaginal rings and ovules. Careful consideration should be given to the patient's health history. Local, low-dose vaginal estrogen is a great option to minimize systemic absorption. If systemic estrogen is considered, remember to add progesterone if the uterus is intact. An oral selective estrogen receptor modifier and a vaginal steroid capsule are available for the treatment of dyspareunia.
Remind the patient it takes time for the treatment to produce results, and a minimum three-month trial is necessary. Cost is always a consideration; encourage the patient to research their coverage for each of the options and educate the office staff so they are knowledgeable resources. A follow-up appointment to review progress and identify any hurdles the woman might be experiencing is highly recommended.
While barriers to treating vulvovaginal atrophy do exist, like with most medical conditions, keeping communication lines open is a key factor for positive patient outcomes.